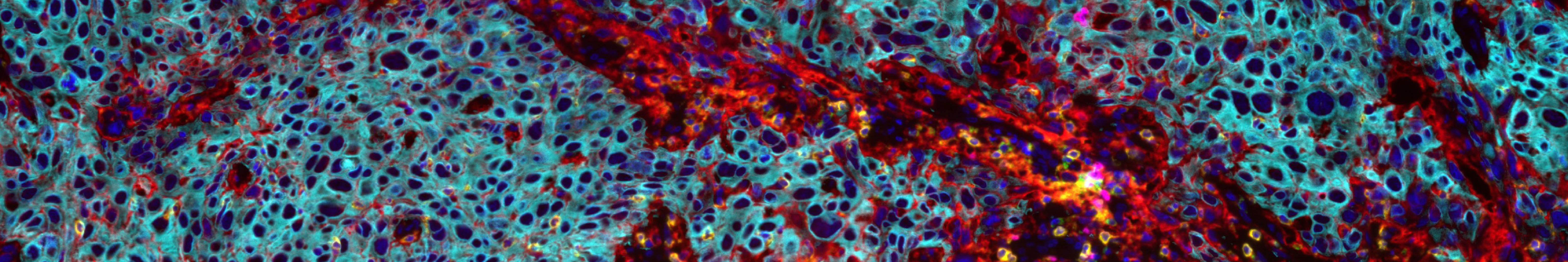
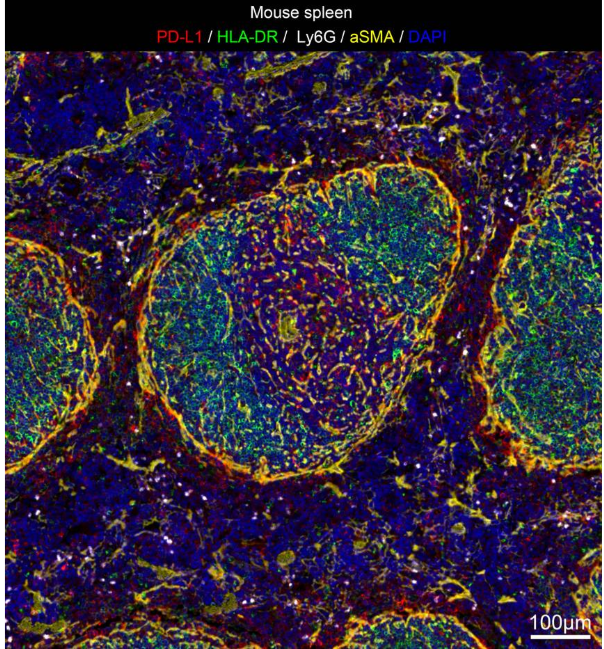
Multiplex Immunohistochemistry (mIHC) technology enables simultaneous labeling and visualization of multiple target proteins on the same tissue section. Combined with high-resolution fluorescence imaging and professional analysis software, it allows in-depth analysis of various biological information in the tissue microenvironment. Typical applications include:
Multiplex Protein Co-localization Analysis: Detecting the co-expression of two or more proteins in the same cell or subcellular structure;
Cell Phenotype Identification and Quantification: Identifying different cell types (such as immune cell typing, tumor cell heterogeneity) through specific marker combinations;
Spatial Relationship Analysis: Evaluating the distribution, proximity relationships, and infiltration patterns of different cell populations in tissues;
Signal Pathway Activity Research: Simultaneously labeling multiple phosphorylated proteins or pathway-related proteins to reveal functional states;
Prognostic Biomarker Discovery: Combining clinical data to quantitatively analyze the correlation between specific cell phenotypes or spatial structures and disease progression.
mIHC is particularly suitable for research fields that require complex microenvironment analysis, such as tumor immunology, neuroscience, and developmental biology.
Tyramide Signal Amplification (TSA) is a highly sensitive detection technology based on enzyme-catalyzed deposition reactions, whose core steps include:
Primary Antibody Binding: Specific primary antibody binding to the target antigen;
Secondary Antibody Conjugation: Horseradish Peroxidase (HRP)-labeled secondary antibody binding to the primary antibody;
Fluorescent Tyramide Activation and Covalent Deposition: Adding fluorescent group-containing tyramide substrate, under the catalysis of HRP and hydrogen peroxide (H₂O₂), tyramide free radicals are rapidly generated and covalently cross-linked to tyrosine residues adjacent to the antigen, achieving local signal amplification;
Antibody Stripping and Cyclic Staining: Removing bound antibodies through mild heating or chemical treatment, retaining covalently bound fluorescent signals, followed by the next round of staining.
TSA technology has high signal-to-noise ratio and signal amplification capabilities, and is not limited by primary antibody species. Only by replacing fluorescent tyramide can multiple rounds of labeling be achieved, making it an ideal method for implementing multicolor mIHC panels.
To ensure the reliability and reproducibility of final experimental results, we recommend conducting systematic pre-experiment verification according to the following steps:
Primary Antibody Specificity Verification:
Perform gradient concentration tests on each antibody in the panel to evaluate its specific signal and background noise;
Multiplex Staining Feasibility Assessment:
Evaluate tissue tolerance based on the tissue type (such as paraffin sections, frozen sections) and processing methods used;
Optimize experimental procedures, such as antigen retrieval conditions, antibody incubation time, etc., to ensure complete tissue morphology and clear signals after multiplex staining.
To ensure the accuracy and reproducibility of Multiplex Immunohistochemistry (mIHC) experimental results, qualified sample preparation is the primary foundation. Below are the key preparation and preservation specifications for three commonly used sample types: paraffin sections, frozen sections, and cell climbing slides.
Paraffin Sections
Preparation Standards: Sections should maintain complete tissue morphology, uniform thickness (usually recommended 3~5 μm), no wrinkles, tears, or knife marks.
Storage Conditions: Can be stored in a dark, dry room temperature environment (21–25°C) for a short period (recommended no more than 2 weeks); for long-term storage, should be sealed and refrigerated at 2–8°C, and attention should be paid to moisture and mold prevention.
Frozen Sections
Pretreatment: Tissues need to be fully fixed with 4% paraformaldehyde first, then dehydrated through gradient sucrose solution (such as 10%~30%) to reduce ice crystal formation, and finally embedded using OCT compound.
Sectioning and Storage: Section thickness is usually 8 μm, also need to use anti-slip slides for attachment to ensure complete and flat tissue. After sectioning, should be quickly sealed and stored in a -80°C low-temperature environment, avoiding repeated freeze-thaw.
Cell Climbing Slides
Cell State: It is recommended to use cells with good adherence and active growth state, with seeding density of 80%–85% being appropriate, avoiding excessive fusion affecting cell morphology and antigen accessibility. Strict aseptic operation is required throughout the culture process to ensure no microbial contamination or abnormal cell aggregation.
Climbing Slide Processing and Storage: Cell climbing slides usually need to be fully washed after fixation (such as 4% paraformaldehyde). If not stained immediately, can be sealed and stored at 2–8°C for a short period; for long-term storage, it is recommended to store at -80°C and avoid freeze-thaw cycles.
Successful mIHC experiments require comprehensive optimization of multiple links, with key attention recommended to the following matters:
Sample Processing:
Keep sections moist throughout to avoid non-specific binding caused by drying;
Choose appropriate antigen retrieval solution and retrieval conditions according to tissue type and target characteristics.
Experimental Design:
Staining Order: Low-abundance targets are labeled first; high-abundance targets are labeled later, reasonably planning fluorescent channel order to avoid crosstalk between adjacent channels, and performing spectral splitting verification and compensation if necessary;
Avoiding Occupancy Effect: Follow the principle of "first low then high" and "first few then many" to avoid excessive occupancy of high-abundance targets in the early stage affecting subsequent staining.
Signal Balance:
Adjust antibody concentration and TSA reaction time through pre-experiments to make each channel's signal intensity moderate and hierarchical, avoiding background increase or signal saturation caused by excessive amplification.
Antibodies:
Transportation Conditions: 4°C cold chain transportation;
Short-term Storage: Can be stored at 4°C for 1–2 weeks after receipt;
Long-term Storage: Recommended to aliquot and store at -20°C, avoiding repeated freeze-thaw;
Validity Period: One year warranty after leaving the warehouse.
TSA Kits:
Storage Conditions: Store in a dark place at 4°C, strictly prevent contamination after opening;
Validity Period: 6 months;
Stability Indicator: If precipitation or significant color change occurs in the fluorescent tyramide solution, it indicates potential failure, and use is not recommended.
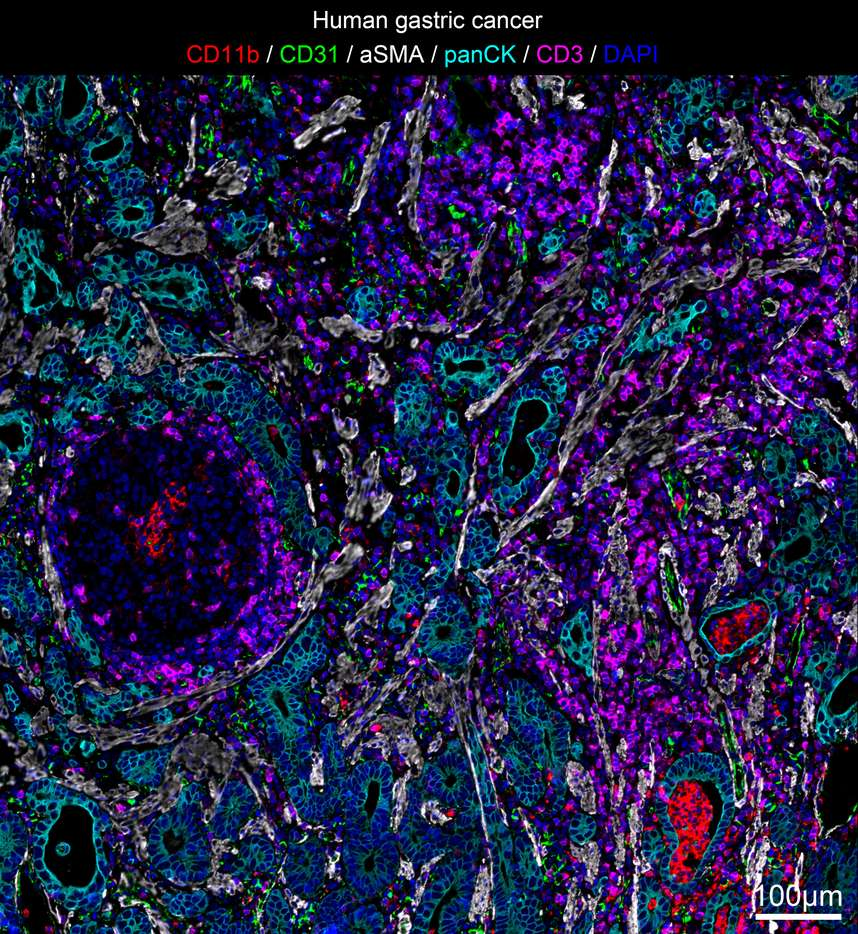
Mature Panels
| Human Gastric Cancer Microenvironment Antibody Panel |
| Red/CD11b, Green/CD31, White/aSMA, Cyan/CD3, Cyan-green/pan CK, Blue/DAPI |
| Labeled Cells<br>T cells (CD3+), myeloid cells (CD11b+), vascular endothelial cells (CD31+), fibroblasts (aSMA+), and tumor cells (pan CK) in the tumor microenvironment |